First, Welding Shielding Gases Types:
There are two main types of Welding Shielding Gases used to protect weld pool:
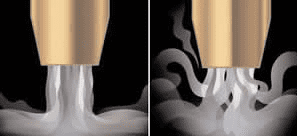
- Inert gases. Inert refers to “non-reactive gases which don’t have valance electron cell thus not taking part into any chemical reactions”. For example, argon as inert gas will not react with any oxygen or metal in the welding arc and puddle, thus we will not see any slag or silicate or oxides on the surface of TIG welding with argon or helium.
- Reactive gases (Active Shielding gases). Reactive gases mean the welding gases that can react will molten weld metal to make their oxides. For example, CO2 as shielding gas, break into ‘CO’ and ‘O’ in the welding arc. The oxygen reacts then with silicon/ manganese presents in the weld puddle to create oxides such as Manganese oxide or silicon oxide.
Why are Welding Shielding Gases is Important?
Welding Shielding Gases serve different purposes. The most important is to shield the molten weld pool from adverse reactions to surrounding conditions.
The 2nd important task is to provide arc ionization for good arc stability. Based on Welding Gases uses, they can be used for weld shielding, purging, and trailing in welding.
The main purposes or objectives of Welding Shielding Gases in welding are:
- Shielding: To ensure the weld pool is protected from the adverse chemical reactions of atmospheric gases.
- Purging: Used to protect weld root (mainly in highly reactive metals such as Stainless Steel, Titanium).
- Trailing or Blanketing: Trailing or Blanketing gases are used to protect the solidifying weld metal that can still react with atmospheric gases. It is not a very common practice and is mainly used for welding highly reactive metals such as Titanium & Tantalum etc. Trailing/ Blanketing is extensively used for Titanium welding where the solidified weld can still react with atmospheric oxygen to create titanium oxide.
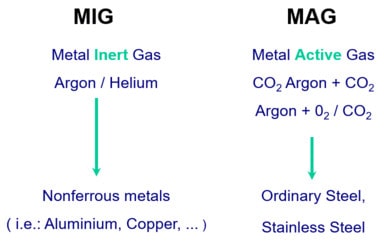
Effects of Welding Shielding Gases in MIG-MAG Welding
| Influence on Weld | Argon+CO2 | Argon+ Oxygen | CO2 |
|---|---|---|---|
| Penetration Depth | Deep & Narrow | Medium | deep & broad |
| Ionization Potential | decreasing with increasing CO2 content | Good | low |
| degree of oxidation | increasing with increasing CO2 content | high as the oxygen content increases, e.g., at 8% O2 | high |
| Porosity | decreasing with increasing CO2 content | most sensitive | very low |
| Gap bridging ability | getting better with decreasing CO2 content | Good | worse than with the mixed gases |
| Spatter formation | increasing with increasing CO2 content | low spatter | Largest spatter ejection, increasing with increasing power |
| Heat input | increasing with increasing CO2 content, Lower cooling rate, lower risk of cracking due to hardening | at the lowest cooling rate high, Greater risk of cracking due to hardening | high Low cooling rate, low risk of cracking due to hardening |
WELDING SHIELDING GASES & THEIR EFFECTS ON WELDING PROPERTIES
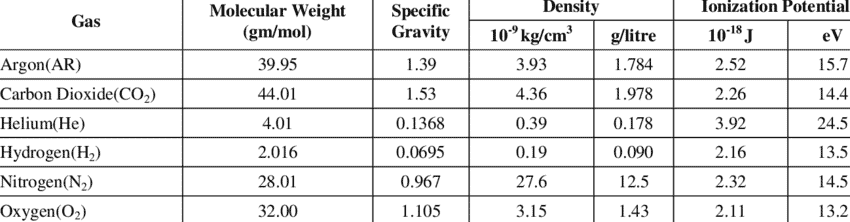
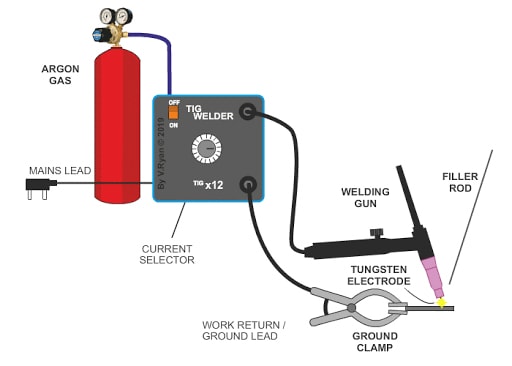
Effect of Argon (Ar) on Welding
Argon as a shielding gas in welding provide deep penetration and less spatters. Argon shielding promoted spray transfer in MIG-MAG Welding.
Argon as an inert gas is an essential shielding & purging gas for reactive metal welding as well as for TIG & Plasma welding operation. Addition of argon with CO2 decrease the shielding gas cost and up to 80% argon offers good quality welds in MIG-MAG & FCAW Welding.
Effect of Helium (He) on Welding
Helium is another inert gas used in Welding operations. Helium has higher lower ionisation rate compare to argon and hence require more welding voltage.
This gives deep and wider penetration profile during welding compare to argon. Pure helium in shielding is beneficial for metals with high thermal conductivity to gives better welding fusion. The density of helium is low so it require higher shielding gas flow for effective shielding blanket during welding.
Cost is another factor with Helium. It is costly compare to argon. It is usually mixed with Argon for TIG welding.
Effect of Hydrogen (H) on Welding
Hydrogen is used in mixed condition with other shielding gases and up to maximum 10% limit due to safety factors. Addition of Hydrogen with oxygen increase depth of welding penetration and also the welding speed in automated welding.
Hydrogen addition with argon for stainless steel welds gives cleaner welds and improved weld profile. The mixture of argon, carbon dioxide, and hydrogen also used in welding to improve penetration, weld profile and the arc temperature.
It is not used for aluminum welding as it can cause welding porosity. And, similarly not for welding of carbon steel and low alloy steel where it can hydrogen cracking.
Effect of Nitrogen (N) on Welding
Nitrogen as a shielding gas in welding improve weld penetration and arc stability. Gas mixed with nitrogen can improve the mechanical properties of alloys containing nitrogen and prevent pitting corrosion and nitrogen loss from the metal.
Nitrogen promotes austenitic phases in austenitic and duplex stainless steel welding. It is used in purging for Duplex stainless steel welding due to this reason.
Effect of Oxygen (O) on Welding
Oxygen is not used purely as a shielding gas but it is mixed with other gases. It is added with argon in tig welding of stainless steel welding to improve the fluidity.
Oxygen gives arc stabilization, weld metal fluidity for a better weld profile and improve metal transfer. Oxygen shielding is not to be used for copper, aluminum or magnesium as it can cause oxidation.
Effect of Carbon-dioxide (CO2) on Welding
Carbon dioxide is cheap and suitable for shielding on carbon steel materials. CO2 increase weld penetration and welding speed but cause more welding spatters.
Also, CO2 shielding gas generates more welding fumes and high heat. CO2 being an reactive gas can not be used for shielding purposes in TIG welding. Also, it can not be used for welding of reactive metals such as Aluminum, copper and magnesium as well as Nickel base alloys.
Usually, CO2 is mixed with argon to improve metal transfer mode from globular to spray transfer. Pure CO2 shielding and up to 80% limit of CO2 in argon, gives globular metal transfer mode. In stick welding, burning of coating generates mainly CO2 as shielding gas.
OVERVIEW OF THE SHIELDING GAS GROUPS BASED ON SFA 5.32
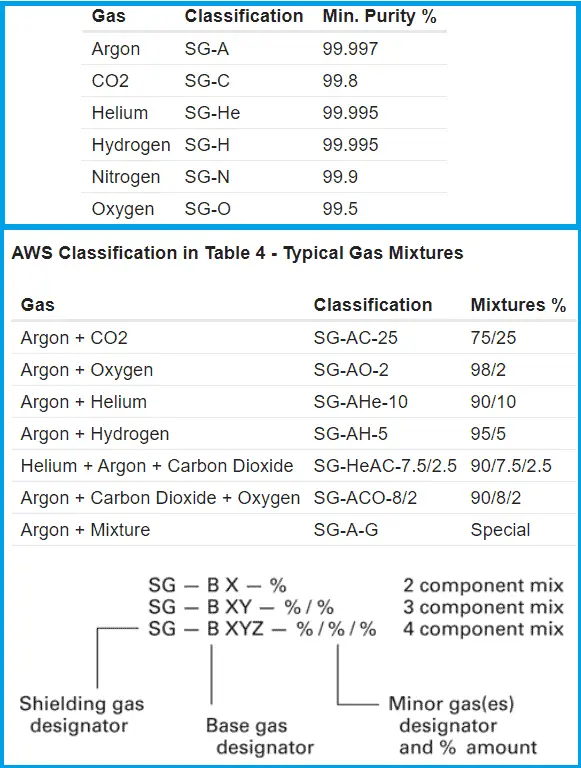
AWS A5.32 “Specification for Welding Shielding Gases”, gives the requirements for the welding shielding gases classification. The Welding gases are classified in AWS A5.32 similar to DIN EN439.
Dew points & Minimum purity level of the shielding gases for TIG, MIG-MAG welding
AWS A5.32 specifies purity and dew point levels that are required for individual gases which are shown in the below table. When you order gas cylinder from gas supplier, and when gas manufacturer supply the gas, he has comply the requirements of AWS A5.32 for gas purity, dew point and mixing ration.
Shielding gases for Aluminum Welding
For gas-shielded metal arc welding of aluminum, both argon and argon helium mixtures are used, whereby argon is the most frequently used shielding gas.
Helium additives lead to higher arc power compared with pure argon and provide for a better weld layout regarding excess weld metal, weld width, and depth or penetration shape. With the same weld penetration, the welding speed can be increased and the risk for the formation of pores and fusing problems can be reduced.
It is important to note that helium, due to its higher ionization potential, leads more or less to a more unstable arc. Therefore 100% helium is only rarely used as a shielding gas and even then mostly in fully mechanized procedures.
Furthermore, to obtain the same protective effect as with argon, the triple amount of gas volume is required because of the lower density of He.
It should be noted that argon gas flow meters are also suitable for setting the helium flow rates if the actual required/set shielding gas amount is determined utilizing correction factors.