What is Hot Cracking in Welding?
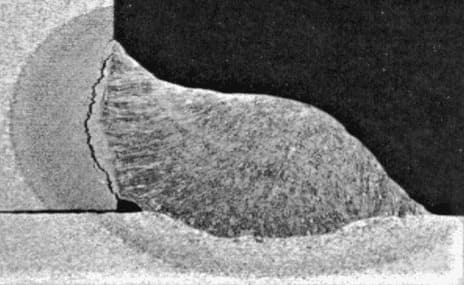
Hot cracking in welding is a type of cracking that can occur in welds during solidification.
It is caused by high shrinkage stresses and can be exacerbated by segregation of low melting phases in the weld center. Hot cracking can lead to weld failure and should be avoided when possible.
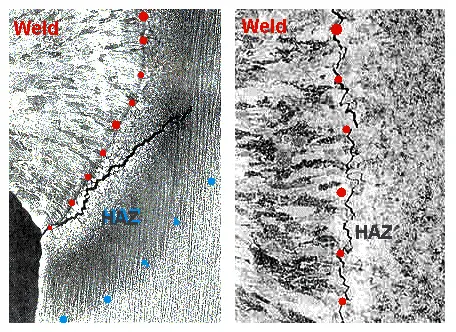
Weldable alloy steels having yield strengths of 56 kg/mm² and higher, sometimes develop hot cracking in the HAZ after welding.
It is important to distinguish this phenomenon from cold cracking, because if it is mistaken for cold cracking and preheat is used as a remedial measure, it might aggravate the overall problem rather than eliminate it.
Hot Cracking Characteristics
Austenitic stainless steel weld-metal is susceptible to minute intergranular cracking called microfissuring, which is not visible to the naked eye.
It is also called hot cracking, because cracking occurs at high temperature as the weld-metal cools from the solidus down to about 980° C.
If the level of stress imposed on the contracting weld-metal is high, the fissures enlarge into visible cracks.
Determining the Susceptibility to Hot Cracking
The susceptibility to hot cracking in welding is determined by:
- (a) the as-deposited weld-metal microstructure,
- (b) the weld-metal composition, especially the content of certain residual or trace elements such as boron, phosphorus, Sulphur, selenium, columbium and tantalum,
- (c) the amount of stress imposed on the weld during cooling,
- (d) the presence of notches.
The first of these factors, namely the weld-metal microstructure determines to a large extent the susceptibility to hot cracking.
Weld-metal, which has a completely austenitic microstructure is highly prone to this defect, while the weld-metal with a duplex structure of delta ferrite in austenite is fairly resistant to it.
As regards weld-metal composition, susceptibility to micro-fissuring can be reduced by a small increase in carbon or nitrogen.
Trace elements which encourage micro-fissuring, in order of their potency, are: boron, phosphorus, sulfur, selenium, silicon, columbium and tantalum.
Since the fully austenitic weld-metal is most susceptible to micro-fissuring during its cooling from the solidus down to approximately 980°C, the amount of stress imposed on the weld-metal must be minimized in this temperature range.
Peening is not effective, because of the difficulty of applying it at the high temperature range.
What Causes Hot Cracking in Welding?
Sulfur is considered the most offensive element in the hot cracking in welding problems.
Carbon and phosphorus lend support to Sulfur in causing hot cracking. Due to the adverse effect of Sulfur on weldability, their content in steel are restricted to a maximum of 0.04%.

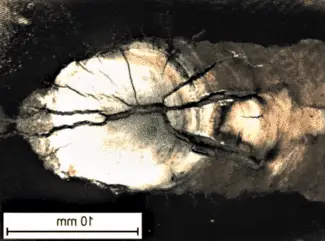
if the Sulfur content of a high-strength alloy steel is near the permissible maximum of 0.04%, it is likely to develop hot cracks in the HAZ when welded under conditions of high restraint.
Hot cracking tests carried out on AISI 4340 steel show that hot cracks under high restraint can be reduced to zero only when the combined S and P contents are below 0.02%-disengass is Carbon increases cracking tendency appreciably.
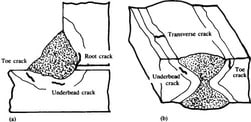
Related Reading: Welding Crack and their Types Overview
How do I stop hot cracking?
Hot cracking in welding is a defect that can occur when there is segregation of low melting phases in the weld center.
There are several ways to prevent hot cracking, including:
- Using a base metal with low Sulfur and phosphorus content.
- Using welding filler material with a higher melting point.
- Welding thick root run to accommodate shrinkage stresses.
- Apply short tack welds in groove before welding full weld.
Effect of Carbon on Hot Cracking
Carbon has been shown to promote inter-dendritic segregation of Sulfur, which increases the likelihood of concentrating lower- melting compounds in grain boundary areas.
Decreasing the carbon content, even by decarburization, reduces the hot cracking in welding tendency.
An annealing treatment which produces spheroidized carbide particles also tends to reduce cracking propensity, apparently because the short austenising time in welding does not dissolve all the globular particles.
Therefore, the actual carbon content in the matrix is lower than the overall carbon analysis for the steel.
Effect of Manganese on Hot Cracking
Manganese has been found to counteract the effect of Sulfur with respect to hot cracking tendency.
This benefit is believed to arise from the strong tendency of manganese to form stable, higher melting-point Sulphides in steel.
Rather than attempt to use large manganese additions as a cure-all, however, good steel makers attempt to reduce hot cracking tendency through reduction of Sulfur content.
Effect of Phosphorus on Hot Cracking
Phosphorus already has been cited as a strong promoter of hot cracking. It appears to act in this manner by increasing the segregation of Sulfur and carbon in the steel-to the point where banding of the Sulphide inclusions and carbide particles can be observed in the microstructure of wrought material.
This banded condition greatly increases the likelihood of hot cracking, and if the banding of carbon is particularly marked, these localized areas of high carbon may be particularly prone to cold cracking as a continuation of the hot cracks.
Effect of Silicon on Hot Cracking
Silicon is believed to cause Sulfur to segregate, and in this manner is likely to increase hot cracking.
Ordinarily, silicon does not have a marked effect on hot cracking susceptibility, because less than 0.50% usually is present in high-strength alloy steels and the content varies over a relatively narrow range.
However, some of the very high strength (heat-treatable) alloy steels make use of silicon contents of about 1%, because this element inhibits softening during tempering in certain temperature ranges.
These steels usually are made with very low sulfur content to avoid undue segregation, which might be promoted by the high silicon content.
Effect of Chromium, molybdenum and vanadium on Hot Cracking
Chromium, molybdenum and vanadium are believed to have a mild beneficial effect upon hot cracking in welding, particularly if a high carbon content is present to aggravate the problem.
These elements possibly tie up some carbon as complex carbides, thus reducing cracking susceptibility by lowering the effective carbon. Nickel exerts an adverse effect upon hot cracking tendency.
Its effect is particularly noticeable when significant sulfur also is present. Presumably, the presence of nickel affects the composition of sulphides formed in the grain boundaries.
Effect of Aluminum on Hot Cracking
Aluminum is another element that seems to influence the composition and distribution of sulphide inclusions.
Residual aluminum in the range of about 0.02/0.06% appears to foster intergranular Sulphide, which noticeably increases cracking susceptibility.
Here we have dealt with the HAZ’s of only carbon steels and low-alloy steels.
It is equally important to know how similar microstructural and related changes take place in the HAZ’s of other industrial metals and alloys as a result of welding, how far they have a deleterious effect on joint properties and serviceability, and how these ill-effects can be controlled.