Welding of Titanium & Filler wire selection guide
Titanium (Ti) is silver-colored metal. It has a strong affinity for oxygen and forms an oxide layer on a clean surface. This leads to natural passivation and provides corrosion resistance to salt or oxidizing acid solution.
Pure Titanium is very ductile and has low strength. A small amount of Aluminum, Oxygen and Nitrogen in the alpha phase increases its strength. It has low thermal expansion and conductivity which improves its weldability.
Material availability & Features:
Titanium has the following four main types:
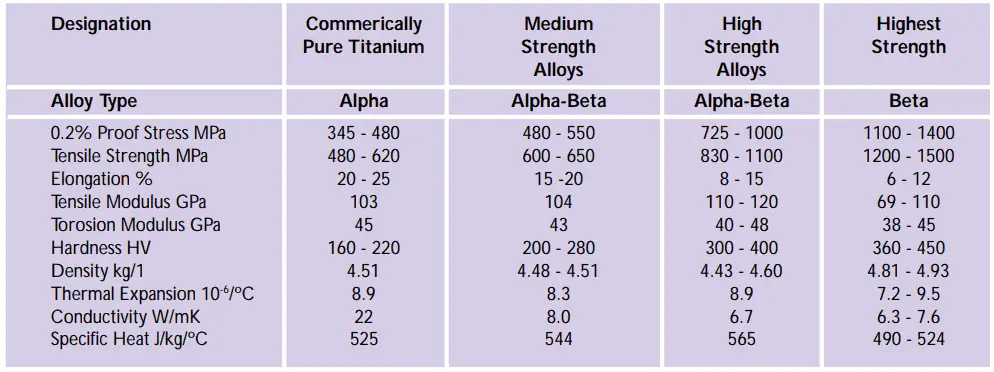
- Pure titanium alloy called CP (without alloying): Common grades are ASTM Gr. 1, 2, 3 & 4.
- Alpha titanium alloy,
- Alpha-beta titanium alloy: Example – Grade 5 (Ti-6Al-4V)
- Beta titanium alloy.
Titanium has the particular advantages of high strength to weight ratio and good corrosion resistance. These have dictated its major application areas Aircraft – because of the strength to weight ratio.
Watch this short interactive video on Titanium Welding Guide:
Chemical plant – because of its high corrosion resistance Titanium, like iron, has two allotropes, i.e. it can exist in two different forms.
It will be remembered that iron had a body-centered cubic crystal structure at room temperature, called ferrite, and on heating, this changed to a face-centered cubic form, called austenite.
It will also be remembered that the body-centered cubic form returned on further heating.
Titanium has a close-packed hexagonal structure, called alpha phase, at room temperature and this changes to a body-centered cubic form, called beta phase, above about 883°C.
The alloys solidify as beta, and under slow cooling, this will transfer to alpha phase by nucleation and growth. Fast cooling gives a meta-stable alpha phase.
This reaction is similar to the martensite reaction in steel but does not generate the same range of desirable properties that the steel martensites and tempered martensites give.
The commercial titanium alloys are either alpha phase, which may or may not be strengthened by solid solution alloying elements, or an alpha phase in a beta matrix where the beta matrix has been retained by a combination of ‘beta stabilizing’ alloying elements and heat treatment.
The principal alloying elements are aluminum and vanadium. Aluminum gives solid solution strengthening and enhances the corrosion resistance of the normal titanium oxide film.
Vanadium is a beta stabilizer and widens the temperature range and cooling conditions where beta can be retained Oxygen is also used as an alloying element.
Oxygen is normally regarded as a harmful impurity but by carefully controlling the oxygen level, it can be used to increase the strength of the titanium without unduly lowering its toughness.
Hydrogen is more harmful. Hydrogen can be absorbed at room temperature from pickling baths or from hydrogen liberated at the cathode if titanium forms a galvanic couple, say with carbon steel. In this case, the carbon steel will corrode and the cathodic reaction, ie the one occurring on the titanium, is the liberation of hydrogen.
Weldability concerns
- Embrittlement is the main issue with titanium welding and hence it must be protected from oxidation during welding.
- Above 400 °C its oxidation resistance decreases rapidly. Therefore, it should be shielded by inert gas (Argon) to avoid contamination from OXYGEN and NITROGEN.
- Oxygen, Nitrogen, and Hydrogen in the form of impurities get picked up by titanium during welding if the weld pool is not protected from the atmosphere. Hydrogen solubility is increased with rising in metal temp. e.g. 8% at 300°C. Therefore, it is essential to give Argon shielding on the weld pool and also for trailing. Hydrogen and Oxygen inclusion in weld reduce the toughness.
- Hydrogen and Oxygen are picked up from moisture from the surrounding. Residual oil, a cleaning agent adhered to the surface causes Hydrogen and carbon pick up.
- Titanium can not be fusion welded directly to Stainless steel, Carbon steel, and Aluminum alloys due to the formation of brittle compounds with these alloys.

Cleaning of Titanium before welding
- Prior to welding the component should be cleaned and dried thoroughly.
- Oil, Fingerprints, grease, paint /dye should be cleaned.
- Chloride and cleaning residues (Residues from Cleaning Agent) on titanium can lead to stress corrosion cracking when it is heated to above 300°C during welding. So, it is essential to clean thoroughly.
- Ordinary Tap water should not be used for rinsing the Titanium parts. De-mineralized (DM) water should be used.
- Before welding, light oxide coating on the weld edges should be removed by Pickling in an aqueous solution of 2-4 % Hydrofluoric acid and 30-40% Nitric acid Followed by DM water rinsing and Drying.
- After cleaning parts should be handled with Lint Free gloves even during welding.
- Any mechanical operation should be followed by pickling to ensure complete removal of scale or any contamination.
- To control porosity in welding, the edges should be scraped withdraw filing, wire brushing. This is required to remove entrapped dirt, small cracks.
- This cleaning is to be carried just before the start of welding. Wherever extended storage is required, it is necessary to store the parts in sealed bags containing silica gel or to be kept in a humidity-controlled room.
- Fixtures used for welding shall need the same cleaning process as Titanium parts.
- It is preferable to have a separate area to set aside for titanium fabrication.
- Water/moisture is a potential source of oxygen and hydrogen and all equipment, jigs, fixtures, etc. should be free from moisture.
- Flexible welding chambers or enclosures can be used for the welding of small components and project sites where possible.
Preparation of the joint for welding
Correct preparation of the weld joint is essential for arc welding of Ti alloys.
- A square-cut edge can be used for all butt and corner welds for thickness upto1 mm.
- Thicker sheets and tubes should be provided with a single V preparation with a 90° included angle and 0-0.5mm root face. This is essential to achieve consistent penetration during root pass welding.
- Acid pickling of weld edges and weld coupons can be used to remove oxygen-contaminated metal from the surface of the titanium.
- The surface of the weld preparation and adjoining metal is critical to the quality of the joint and should be scrupulously cleaned prior to welding.
- The surface should be inspected to see whether a final polishing operation is necessary. The smoothness of weld edges is important for reduced porosity in arc welding.
Protection during welding of Titanium alloys & welding consideration for Titanium Welding
- Due to the sensitivity of Titanium to embrittlement by Oxygen, Hydrogen & Nitrogen, the entire weldment and its surrounding (Around 25 mm all round) should not be allowed to remain above 300°C and this area should be protected by Argon Trailing during welding till it cooled below 300°C as best practice.
- A Trailing shield is required to use for welding Titanium grades. The shield from the gas cup may not be adequate, therefore a trailing shield is to be fitted to the welding torch to provide extra Argon which keeps the hot weld zone shielded with Argon longer than if it were just with a torch nozzle alone. This extra shielding gives time for the metal to cool below the temperature at which it oxidizes.

- High purity Argon gas must be used for welding Titanium. Recommended purity of Argon is 99.999%.
- It is recommended to check the purity of Argon gas for each batch of cylinders. Therefore, it is recommended to weld a piece of the plate prior to production weld and check that piece for bend (Bend dia. -8 t) & Visually for Porosity. (Oxygen reduces the ductility).
- Care to be taken to ensure that moisture and air are not leaked (through the faulty system –i.e. Regulators, Pipe connections, etc.).
- Turning, Milling, Planning- These are the most popular methods used for edge preparation. Care should be taken that material is not overheated during machining.
Discoloration in Titanium
The color of the weld is often used as a measure of contamination level in Titanium Welding.
- A Silvery shiny color is indicative of correct shielding & it is desired to have this color. Satisfactory weld.
- Light & dark bronze/brown color indicates a very small amount of contamination.
- Light & Dark Blue color indicates the heavy amount of contamination. NOT Acceptable.
- Grey-blue, Grey, and White color indicate very heavy contamination. NOT Acceptable.
- When several passes are to be deposited in groove weld, no contamination in between passes/layers is acceptable.
- Discoloration occurs during welding and not always sign of poor shielding. Indeed, dark brown ‘tramlines’ parallel to the weld bead is commonly encountered in fusion welding.
- Moisture comes from improper cleaning and drying of joint prior to welding
- Improper tack welding & wide root opening. Hardness testing can be used to provide supporting evidence for contamination as an alternative to color criteria since contaminated welds will exhibit higher hardness.

Porosity in Ti weldment is caused due to the following:
- Moisture which comes from improper cleaning and drying of joint prior to welding
- Improper tack welding & wide root opening.
- Porosity increases with a decrease in welding speed.
TIG Welding techniques:
- TIG welding power source should be equipped with a non-contact arc strike to prevent any tungsten contamination of the weld which occurs if a touch-down technique is used. Should the tungsten electrode touch the weld then both must be carefully examined before restarting. Any tungsten in the weld must be excavated.
- The power source should also be capable of breaking the arc on completion of a weld run without stopping the inert gas flow.
- Bigger size of the gas nozzle to be used to ensure the adequate shielding of the weld pool.
- Pre-flow of 6-10 seconds and post-flow of 25-30 seconds is recommended.
- A stringer bead technique is recommended for Ti welding.
- Heat input to be kept to the minimum possible and never more than 1.0 KJ/mm.
- No preheating is required for Ti welding.
Preheating and Interpass temperature for Titanium welding
- Maximum Interpass temperature should be kept as low as possible and in no case can be more than 80°C.
- Staggered welding technique to be followed to avoid localized overheating of the weld zone.
Shielding & purging gases for Titanium welding
Titanium is a reactive metal, making it an element that reacts with oxygen readily to make Titanium oxide at even room temperature. The Titanium oxide layer is having a high melting temperature than the base metal, so it must be cleaned before welding.
If titanium is heated it converts to highly reactive material that easily combines with other elements/ gases such as oxygen, hydrogen, and carbon to form their oxides.
This oxide formation makes titanium brittle and hence protection from oxide formation must be applied when welding. Due to this reason, the weld & HAZ is shielded using inert gas shielding till the temperature decreases below 800°F. For welding:
- Use clean and non-volatile piping to supply gas for purging and shielding.
- Shielding gases must be free from other gases such as Nitrogen, oxygen, carbon dioxide, or hydrogen gas that can cause oxide formation.
- All gases used for shielding or purging shall have a dew point of -60°F (-51 °C) or better and oxygen content shall not exceed 50 ppm.

Where titanium is used?
Titanium is renowned for its strength and its durability, making it one of the most sought-after materials in modern manufacturing. Its use has spread across a wide range of industries, from aerospace applications to medical implants. This versatile metal offers manufacturers a reliable and cost-effective solution for many components and products.
It is widely used in the aerospace industry due to its lightweight and strong properties; titanium components are often found in aircraft engines and airframes.
The automotive industry also uses titanium extensively in engine components such as exhaust systems, valvetrain parts, gaskets, spark plugs and connecting rods.
Titanium is also popular for decorative purposes; jewellery makers often choose it for rings, necklaces and other items due to its natural beauty and resilience.









