What is Copper Sulfate Test?
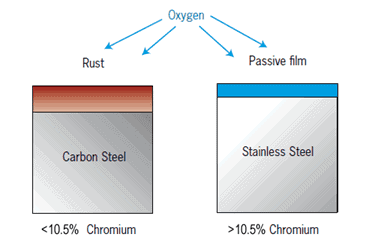
Copper sulfate test is a chemical test carried out to find the presence of Iron (Fe) in stainless steel by visual verification. The test works on the principle of oxidation-reduction chemical reaction that causes the dissolved copper ions to settle or stick to the iron present on the surface.
Copper sulfate solution is applied by swabbing or brushing to the surface of interest and if any iron particles are presents on the surface they will react with the solution and change to brownish color.

This highly sensitive technique is advised for the presence of iron or iron oxide on the surface of austenitic 200 series and 300 Series, duplex stainless steel, the precipitation hardening alloys, and the ferritic 400 Series stainless steel containing 16 % chromium or higher.
The test is not advised for the martensitic stainless steel and low chromium ferritic stainless steels of the 400 types since the test may show a +ve reaction on these materials.
Copper Sulfate Standard
The copper sulfate test is defined in the ASTM A 380, ISO 15730 standard & MIL-STD-753 (US Military standard).
Materials for Copper Sulfate test
Copper Sulfate test is used for the following materials:
- SS200 series,
- S300 Series
- Duplex stainless steel
- Precipitation hardening alloys
- Ferritic Stainless steel, 400 series having more than 16% chromium
During welding of stainless steel components, iron particles can stick to the stainless steel parts and could be the initiation point for corrosion, here a copper sulfate test can be used that no iron (Fe) is available on the stainless steel surface.
Or when a carbon steel component is cladded with stainless steel or Inconel alloy and to verify it is free from iron, a copper sulfate test can be used.
Preparing the Copper Sulfate Solution

The test solution is prepared by first adding sulfuric acid to distilled water and then dissolving copper sulfate in the following proportion.
Distilled water: 250 mL
95–100 % Sulfuric acid (H2SO4) :1 mL
Copper sulfate pentahydrate (CuSO4·5H2O): 4 g
Copper Sulphate Test Procedure: Steps for Copper Sulfate test
Copper Sulphate Test Procedure is used to detect presence of free iron on material surface. The main steps for Copper Sulphate Test Procedure are:
- Prepare the test solution and mix the chemicals with water in quantity given above. Let it to mix for 4 to 6 minutes. Never use solution which is prepared more than 2 weeks before.
- Apply the test solution to the surface to be inspected by swabbing or brushing, apply additional solution if needed to keep the surface wet during the test duration for six minutes.
- Rinse the surface fully and take care not to disturb copper deposit if any till the part is completely dried.
- Perform the visual inspection for the presence of any copper deposit on the area of test. Any copper deposit indicates the presence of free iron.
Test Temperature
Normally performed at Room Temperature since the copper sulphate solution contains Water, it will evaporate at high temperatures. Also during any Repair welding, after removal of metal wait for the temperature to come down.
Drying Method
After completion washed with 30 ppm water and cleaned with a LINT free cloth and further Cleaned with a Solvent such as Acetone.
Safety Precautions
a. While preparing the solution, since Acid is involved proper suitable Gloves and Eye
protection to be used.
b. During disposal keep away from fumes, use right PPE and dispose as per the regulations.
Please check the color after application of copper sulfate solution. Rust is brownish in color.
When copper sullfate deposit’s over contaminate iron it will be bullish.
Test Result Interpretation
The copper sulfate test is a qualitative test method. The results are interpreted as pass or fail by visual inspection without the aid of any other inspection tool.
The test principle uses the oxidation by chemical reaction of iron particles if any presents and turning them into copper color as visible in the below picture.
The test will not tell how much iron is present on the surface but gives an indication in a qualitative manner that iron is certainly presented on the surface being tested.

Copper sulfate test for passivation parts
Copper sulfate test for passivation parts is carried out by applying the copper sulfate solution as described above- Steps for copper sulfate test.
ASTM Copper Sulfate Test
ASTM Copper Sulfate Test is specified in ASTM A 380 Standard.
Copper sulfate test for Clad material removal inspection
Copper sulfate test is also used for the repair of clad and weld overlay applications to ensure the full removal of the clad layer before the start of welding on the carbon steel material.
-For clad restoration or clad removal due to wrong welding clad filler, after the removal of clad, the area shall be etched by nitric acid or copper sulfate solution to confirm the removal of clad material.
– Before the back cladding butt joints, apply the right precautions to avoid cross-contaminating the cladding and backing material weld metal.
The location of weld cladding will not be visible after grinding but etching the area with copper sulfate solution or nitric acid is useful to confirm the weld location and base material.
Related Reading: What is Passivation & Pickling Process?
Copper Sulfate Test for Passivation
Copper sulfate test is used to determine whether a metal has been properly passivated. The test can be used as a quality test to decide if further passivation is required or not.
Copper Sulfate Test for Passivation check for the presence of free iron on the material surface. The test is carried out at the ambient temperature conditions.
Copper Sulfate Test for Passivation Procedure
- Apply the copper sulfate solution on the material surface for approximate 5 minute.
- After 5 minute, rinse & dry the part completely carefully. If any copper deposit present, it can be noticed visually.
- The presence of pinkish/ brown color shows that free iron is still on the material surface and it require further passivation.
- The test is acceptable if there is no copper deposit is present.
Sources Of Iron contamination
- Stainless steel in contact with carbon steel or LAS.
- Working on carbon steel near to Stainless steel materials. Example, Welding or Grinding of
carbon steel materials which impinges on Stainless steel. - Use of grinding wheels, Buffing discs etc.. on carbon steel which further used on stainless steel
materials. - Open yard storage of Stainless steel materials being prone to iron contamination from the
atmosphere. - In a SS Clad which had been stripped back 5-10mm and carry out welding & grinding of carbon
steel portion resulting in spatters and grinding sparks falling on clad SS portions. - Contamination from Handling materials either previously used on carbon steel or containing
carbon steel parts coming in contact with SS materials.
Effects of Iron contamination
- Embedded iron particles in the SS materials may react with oxygen and moisture and forms Iron
oxide or rust which may deter the passivated layer of SS materials. - SS materials may start to PIT due to the reaction of embedded Iron particles with atmosphere.
Pits are considered very dangerous in SS materials as it may eventually result in Pitting
corrosion.
Prevention of Iron Contamination during Fabrication
- Use Separate shops for Fabrication of Carbon steel materials and SS materials.
- Use tools dedicated to SS materials free of carbon steel (can be checked by a Magnet).
- Avoid open yard storage of SS materials and if unavoidable cover the SS materials with proper
cover or Cloth such as Tarpalene. - While welding or Grinding Carbon steel portion of SS clad materials, cover the Clad portion with
proper cover - Handling materials getting in contact with SS materials shall be covered with a strip of SS
materials and ensure no carbon steel gets in contact with SS materials. - Moreover Ropes, strips, slings during handling must be ensured that they are not used on
Carbon steel materials when handling SS Materials.