What is the Coefficient of Thermal expansion or CET?
The coefficient of thermal expansion is an estimation (change in size over a defined size at a certain temperature) of how much a metal expands when heating and contracts during cooling based on a specific temperature.
Materials having a higher coefficient of thermal expansion will be having more expansion & contraction during heating & cooling while those with lower CET values, will expand & contract least.
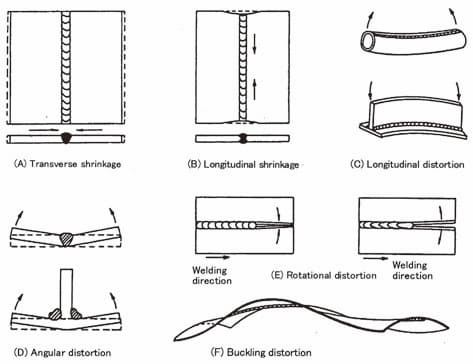
In welding fabrication, CET is directly responsible for distortion because the metal having higher CET values are prone to higher dimension instability and result in distortion very frequently.

It is a physical property of metals. It measures how certain materials will behave when they are heated.
These changes are usually changes in sizes i.e. increase in size. Due to different atomic structure arrangements, different materials behave in different manners once they are heated.
This change in expansion is measured by the coefficient of thermal expansion. The unit of thermal expansion is µm/m · K, or 10–6/K.
Understanding the Coefficient of Thermal Expansion
Metals when subjected to heat expand and shrink when subjected to cold and vice-versa. The phenomenon where metal exhibits this special feature is known as the Coefficient of Thermal Expansion of materials.
These properties related to thermal energy for material is very valuable for engineers & scientist. Here, in this post, we are limiting our discussion only related to Welding Engineering.
This property (α1= Coefficient of Thermal Expansion) can be shown by a mathematical formula as given below:
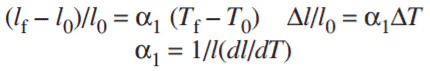
Here,l0 is the initial length, lf is the final length, T0– is the Initial temperature and Tf is the final temperature.
How To Measure CTE or Thermal Expansion Coefficient
Measurement of thermal expansion coefficient (CTE) requires measuring the displacement & temperature characteristics.
This is executed by a given material specimen by subjecting it to heat. The subsequential physical changes that arise due to the applied thermal cycle are evaluated by considering the change in dimensions and temperature to find out the CTE values for the subjected material.
Below are the methods used for the measurements of the coefficient of thermal expansion (CTE) for a metal:
- Interferometry
- Dilatometry,
- Thermomechanical analysis.
Coefficient of Thermal Expansion in Steels
Different materials due to their microstructural arrangement exhibit varied thermal expansion when subjected to heat.
With most of the engineering materials related to welding are carbon steel, chromium steel (ferritic stainless steel) and austenitic stainless steel.
Simple ferritic stainless steel have a coefficient comparable to carbon steel, but for austenitic stainless steel material is almost 12 times higher. The property is very useful when performing corrosion resistance overlay of austenitic base chemistry on the carbon steel material substrate.
Here , the predominately austenitic base cladding will have much higher thermal expansion compared to the carbon steel base metal.
Due to this reason, such clads are not subjected to post weld heat treatment (PWHT). If PWHT is carried out, this will result in a broad difference of co-efficient of thermal expansion between carbon steel and austenitic stainless steel metallurgy and will have very high resultant residual stresses.
This combined high thermal expansion and lower thermal conductivity for these stainless steel require utmost care to mitigate welding distortion & high residual stresses during welding.
E.g., while welding of Cr-Ni Alloys (SS304L, SS316L) it is recommended to use lower welding heat, stringer weld beads, and higher travel speed.
Coefficient of Thermal Expansion in Welding
Welding is process that induces thermal energy in the materials. This thermal energy is directly responsible for change in the materials dimensions due to resultant coefficient of thermal expansion.
E.g. ,In case of similar metallurgy, the resultant expansion is balances while in case of dissimilar material welding, CTE plays an important role.
Higher variations in the coefficient of thermal expansion values of nearby materials during cooling shall create tensile & compressive stresses.
The material facing this tensile stress can get a hot-crack during welding, or can cold crack during service if these residual stresses are not relieved. an example of such a case is in refineries where weld of carbon steel and copper alloy are used together.
The most common problem in dissimilar metals welding (DMW) is because of the differences in the physical properties (mainly the coefficient of thermal expansion and heat conductivity) of the base materials as well as on certain levels of metallurgical incompatibility.
The weld thermal cycle produces a differently featured heat-affected zone (HAZ), associated by microstructural changes in the material microstructure, and these changes can drastically change the acceptance of the welding joint such as meeting hardness for NACE in carbon & low alloy steel or fulfilling the phases requirements for duplex stainless steel welding.
Coefficient of thermal expansion for metal
The coefficient of thermal expansion of mostly used materials in the welding is given in the below table.
For the coefficient of all materials. The coefficient of thermal expansion for carbon steel is 6.5- 10-6/°F, and austenitic stainless steel is 6.4 to 14-10-6/°F.
| Material | CTE (10-6/K ) | CTE (10-6/°F) |
| PureTungsten (W) | 4.5–4.6 | 2.5–2.6 |
| Iron-cobalt-nickel alloys | 0.6–8.7 | 0.3–4.8 |
| Pure Chromium (Cr) | 4.9–8.2 | 2.7–4.6 |
| Pure Titanium (Ti) | 8.4–8.6 | 4.7–4.8 |
| Molybdenum alloys | 4.0–14 | 2.2–7.8 |
| Ferritic stainless steel | 9.3–12 | 5.2–6.5 |
| Cast ferritic stainless steel | 11 | 5.9 |
| Martensitic stainless steel | 9.5–12 | 5.3–6.6 |
| Iron carbon alloys | 10–12 | 5.6–6.5 |
| Wrought iron | 11 | 6.4 |
| Structural steel | 12 | 6.5 |
| Nickel chromium molybdenum alloy steel | 10–13 | 5.7–7.3 |
| High-manganese carbon steel | 11–13 | 6.2–7.0 |
| Malleable cast iron | 10–14 | 5.6–7.6 |
| Ductile medium-silicon cast iron | 11–14 | 6.0–7.5 |
| Gray cast iron | 11–15 | 6.0–8.5 |
| Austenitic stainless steel | 9.8–25 | 5.4–14 |
Expansion of Metals on Heating
Heating metals can cause them to expand. This expansion can cause problems, such as the metal breaking or tearing something it is attached to.
Heating causes atoms in the metal to vibrate more, and these vibrations cause the metal to expand. The amount of expansion depends on the metal and how hot it is heated. Some metals, such as aluminum, expand a lot when heated, while others, such as steel, expand very little.
Practically, when metal is heated, the atoms within it become more active. They move around more and take up more space. This is because the increase in kinetic energy of the atoms causes them to spread out. The metal will expand as a result.
Does Metal Expand in Cold?
Metal does not actually expand when the temperature goes down. In fact, metal shrinks when the temperature goes down.
This is due to the metal’s molecular structure and the way that atoms are packed together. When the temperature decreases, the atoms contract and the metal becomes smaller.
Metal does not expand when it is in a colder environment. In fact, it contracts. This is why bridges and other metal structures are able to withstand cold weather; if they expanded, the stress on the metal would be too much and it would eventually break.
There are a few metals that do expand slightly in cold weather, but this expansion is negligible and does not cause any problems.
Coefficient of Thermal Expansion of Steel
Steel is a material that has a relatively high coefficient of thermal expansion. This means that the steel will expand when it is heated and contract when it is cooled.
The value of the coefficient of thermal expansion for steel can vary depending on the type of steel and the manufacturing process used. In general, the value for steel is around 5.6–6.5 x 10-6 per degree Fahrenheit.
Which metal expands the most when heated?
The answer to this question is not as straightforward as one might think. Metals expand at different rates when heated, and the amount of expansion can vary based on the temperature change and the metal’s composition.
Pure metals, like gold and silver, typically expand the most when heated. Alloys, or metals that are composed of two or more elements, often expand less than pure metals. For example, steel expands much less than gold when heated.
There are a few factors that can influence a metal’s rate of expansion. The temperature change itself is one factor – a metal that is subjected to a large temperature change will expand more than one that experiences a small change.
The composition of the metal is also important; alloys typically expand less than pure metals because they have a smaller volume coefficient of thermal expansion.