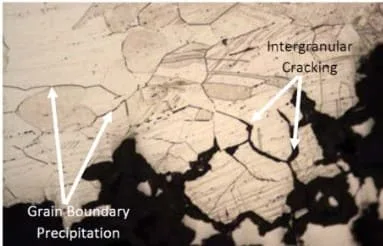
Pitting Corrosion
Pitting corrosion is one of the most aggressive forms of corrosion. It is characterized by small, localized holes or pits in the metal surface.
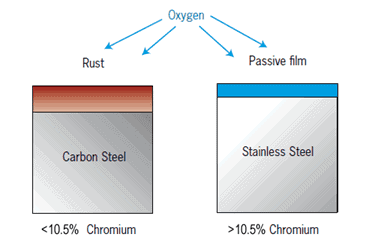
Some metals protect their surface from corrosion by forming an oxide layer. This is the case with all non-ferrous metals, but also with stainless steels, which also form a passive layer of oxides due to their alloying element. In these cases, continuous corrosion can no longer occur – but pitting corrosion occurs.
The pits are usually round or oval-shaped and can be very deep, penetrating well below the surface of the metal. Pitting corrosion is caused by a combination of chemical and electrochemical reactions.
The most common cause is when a metal comes into contact with an acidic solution, such as vinegar or lemon juice, for example. As the acid attacks the metal, it creates small holes or pits in the surface.
There are many corrosive medias that cause pitting corrosion, and they are:
- Water (H2O)
- Sulfides (S2−)
- Chlorides (Cl-)
- Bromides (Br−)
- Fluorides (F−)

Pitting corrosion can also be caused by electrolysis, which is the process of using an electric current to break down water molecules into their component parts:
- Hydrogen and
- Oxygen.
When electrolysis occurs in the presence of a metal, it can create small pits or holes in the surface of the metal.
What is Pitting Corrosion?
Under certain conditions, particularly involving high concentrations of chlorides (such as sodium chloride in sea water), moderately high temperatures and exacerbated by low pH (i.e ., acidic conditions), very localized corrosion can occur leading to perforation of pipes and fittings etc.
This is not related to published corrosion data as it is an extremely localized and severe corrosion which can penetrate right through the cross section of the component. Grades high in chromium, and particularly molybdenum and nitrogen, are more resistant to pitting corrosion.
One reason why pitting corrosion is so serious is that once a pit is initiated there is a strong tendency for it to continue to grow, even although the majority of the surrounding steel is still untouched.
How does Pitting Corrosion occur?
Pitting corrosion is a form of localized corrosion that can occur on metal surfaces such as steel, aluminum, and titanium.
This type of corrosion causes the metal to corrode from the inside out, resulting in tiny pits or holes in the surface of the material.
Pitting corrosion occurs when there is an anodic site and cathodic site present on metals exposed to water or any other electrolyte.
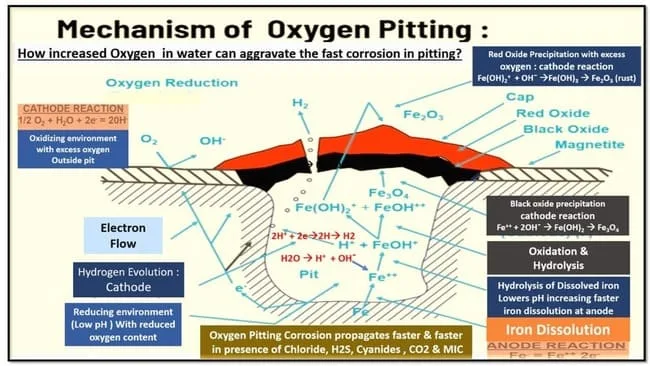
The anodic site provides electrons which cause oxidation at the surface while cathodic sites provide electrons to reduce oxygen at their location leading to a decrease in pH causing pitting corrosion.
Pitting corrosion can be a major problem for industrial companies as it leads to costly repairs and even total replacement of equipment.
Prerequisites for pitting corrosion
Pitting corrosion occurs only in aqueous solutions in which chloride or bromide ions are present. In the case of copper and copper alloys, on the other hand, a high sulphate content is decisive for the occurrence of pitting corrosion.
In addition, some other factors have a beneficial effect:
- low pH of the electrolyte
- high temperature
- low oxygen content in the electrolyte
- low flow velocities
What is Pitting corrosion test?
The tendency for a particular steel to be attacked by pitting corrosion can be evaluated in the laboratory. A number of standard tests have been devised, the most common of which is that given in ASTM G48.
The pitting corrosion test as per ASTM G48, is a standard test method for detecting and measuring pitting corrosion in metals.
Read more: Corrosion Testing Methods and Standards.
The test involves immersing a metal specimen in an electrolyte solution and applying a potential difference between the specimen and a reference electrode.
The current flowing through the solution causes pitting to occur on the specimen’s surface. The depth of the pits is measured to determine the severity of the pitting corrosion.
The ASTM G48 test method covers the procedures for conducting pitting potential measurements to determine the susceptibility of stainless steels and related alloys to pitting and crevice corrosion in oxidizing chloride environments.
This test method is applicable to austenitic, duplex, ferritic, martensitic, precipitation hardening, and other types of stainless steels and related alloys.
How to prevent pitting corrosion?
Pitting corrosion is a type of localized corrosion that can occur in many metals. It is characterized by small holes or pits on the metal surface.
Pitting corrosion can be caused by many factors, including chemicals, electrochemical reactions, and microorganisms.
There are several ways to prevent pitting corrosion. Such as:
- One way is to coat the metal with a protective film. This film can be made of metals, plastics, or other materials.
- Another way to prevent pitting corrosion is to control the environment in which the metal is exposed. This includes controlling the pH, temperature, and chloride ion concentration.
- Finally, regular inspection and maintenance of metal surfaces can help to identify and repair areas of pitting corrosion before they cause significant damage.
Pitting Resistance Equivalent Number (PREN)
PREN, Pitting Resistance Equivalent Number is a measure of a material’s resistance to pitting corrosion.
PREN, Pitting Resistance Equivalent Number is a method of measuring the pitting resistance of stainless steels. The PREN number is calculated by taking into account the chromium (Cr), molybdenum (Mo), Tungsten (W) and nitrogen (N) content of the steel.
Below is the most widely used PREN Formula:
- PREN = %Cr + 3.3 x (%Mo + 0.5%W) + 16 x %N
The PREN values for common stainless steel range from about 12 to 40. The higher the PREN value, the greater the resistance to pitting corrosion. For example, SS410 has a PREN Number of 11.5 while SS316L has a PREN Number of 24.
So, 316L (austenitic stainless steel) has double the pitting resistance compared to SS410 (Ferritic Stainless Steel).
Below chart provides PREN Number for Various stainless materials.
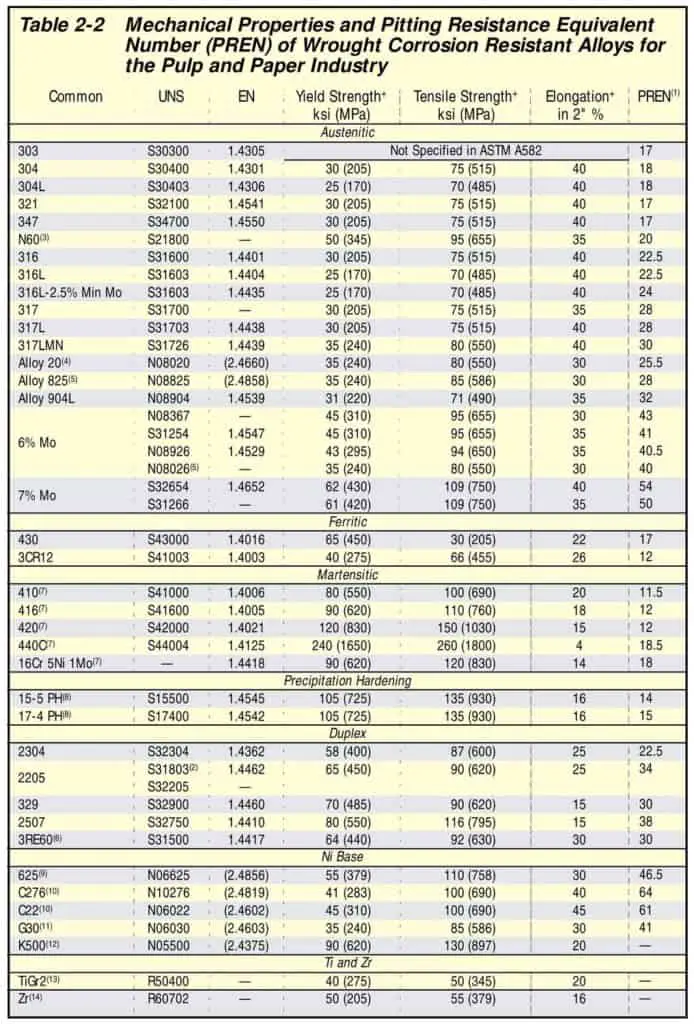
Pitting is a type of corrosion that can occur in stainless steel. It is caused by the formation of small holes in the surface of the steel. Pitting can be prevented by using steel with a high PREN number.
The PREN number is an important factor to consider when choosing stainless steel for an application where pitting resistance is required.